|
4/1/2024 0 Comments Electron affinity trend 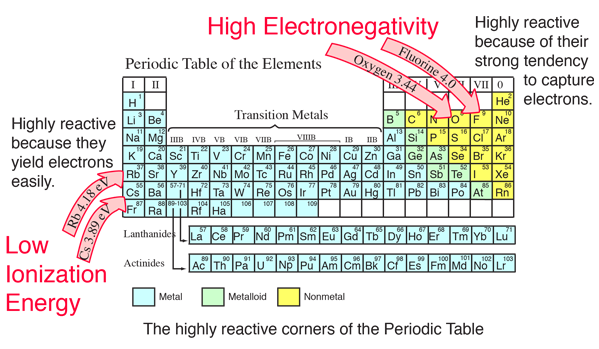
As the number of protons increase, the nucleus of the atom becomes more positively charged. This is because the number of protons increases moving to the right of the row. When moving left to right across a period, the electron affinity increases. Going Left and Right Across Periods (Rows) The new orbital is further away from the nucleus, meaning the attraction between the positively charged nucleus and the new electron decrease. This is because as you go down the period table, new valence shells are added increasing the atomic radius. When moving down a group, the electron affinity generally decreases. The more negative the electron affinity is, the more likely an atom will accept an electron. It measures the energy change in the atom that occurs when an atom gains an electron. Electron affinity measures the ability of an atom to accept an electron. These patterns are called periodic trends. 
The tendencies of these patterns increase or decrease as you move along the columns and rows of the table. Specific patterns of certain elemental characteristics are present in the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
